Tiny Cell Motors Move Like Seesaws
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
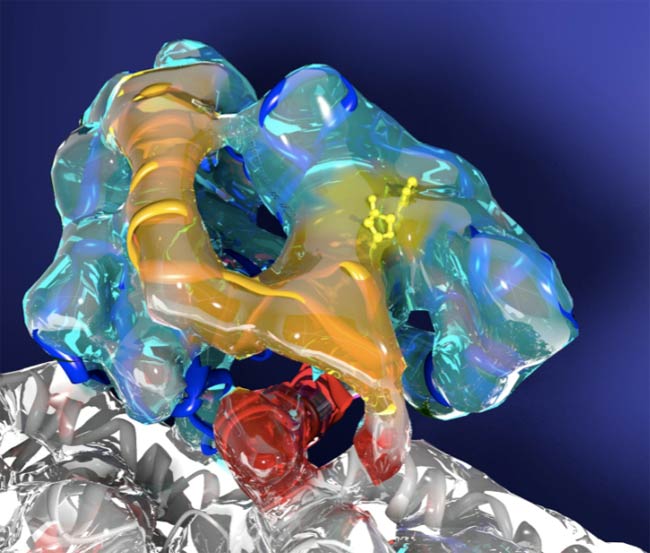
Scientists have zoomed in on the tiny molecular motors inside the body's cells and developed a better picture of how exactly they trek along their cellular tracks. Turns out, part of the motor moves like an atomic-scale seesaw, according to a recent study.
The finding is based on high-resolution images of kinesin, a so-called motor protein that moves cargo inside your cells and also helps cells divide.
A better understanding how kinesin works may help scientist develop new drugs to fight diseases such as cancer, the researchers say. Blocking kinesin movement could prevent cells from dividing and lead to cell death.
How proteins walk
Scientists had known kinesin "walks" along cellular scaffolding known as microtubules, but weren't sure about the precise mechanism.
"We found that there is a pivot point, where the kinesin motor attaches to the microtubule, which acts like a fulcrum and causes kinesin to rock up and down like a seesaw as it moves along the microtubule," said study author Kenneth Downing, a biophysicist at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory, who conducted the work with then postdoctoral fellow Charles Sindelar.
The work provides the most complete picture yet of kinesin's movement, according to Sindelar, now at Brandeis University, since it captures sharp images from all different stages in the motion cycle. The results also give scientists a better idea of how kinesin uses energy to shuttle itself along the scaffolding.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"In a sense you can watch the energy being converted into movement," Sindelar told LiveScience.
Tiny pictures
Studying motion on the atomic scale is no easy task. To see a protein's individual atoms, scientists often use an imaging technique called X-ray crystallography, which requires a protein to be turned into a crystal.
But scientist can't turn kinesin into a crystal if they want to see it "walk." So they used electron microscopy, an imaging technique that magnifies small things (much like a microscope does, but instead of using light, it uses electrons), and allows scientists to study how cellular processes occur in real life. While previous studies have examined kinesin movement with electron microscopy, Downing and Sindelar used a new approach to process the data, one that allowed them to obtain higher resolution, or sharper images.
The resulting pictures showed a detailed outline of the protein and gave the scientists a guide to its movement. But they were not quite close enough to see the individual atoms. To solve this problem, the researchers compared their "walking" pictures with even sharper still images using X-ray crystallography. Together, "these images allow us to build an atomic model that explains the basic function [of kinesin]," Downing said.
The work was published online this week in the journal Proceedings of the National Academy of Sciences.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.

 Live Science Plus
Live Science Plus









