This Bizarre, Overstuffed Atom Is the Turducken of the Microscopic World
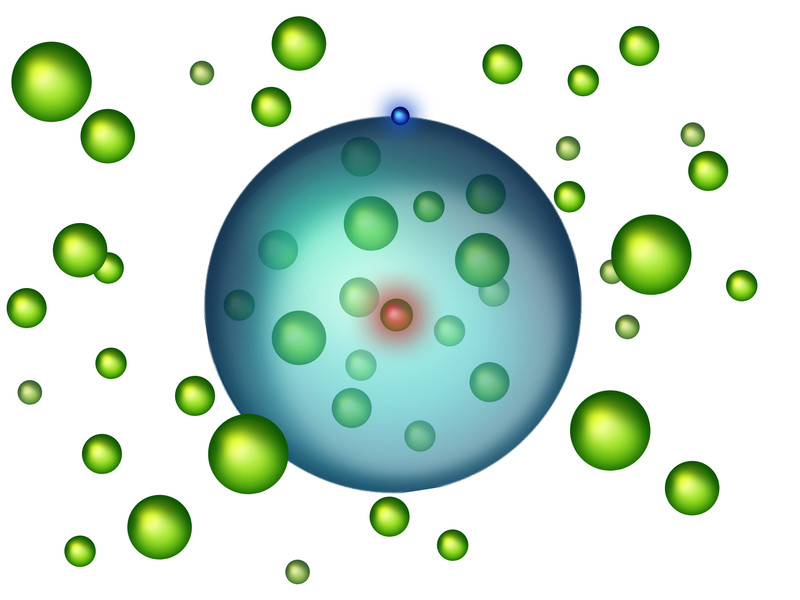
It's the turducken of the microscopic world: an atom crammed with other atoms, linked up with other atoms, to create an exotic new state of matter.
The overstuffed atom uses the special properties of something known as a Rydberg atom. Pump these atoms with lots of extra energy, and the electrons get very excited. As a result, the space between the nucleus and the electron's outer orbit becomes huge, researchers reported in a new study.
"The average distance between the electron and its nucleus can be as large as several hundred nanometers — that is more than 1,000 times the radius of a hydrogen atom," Joachim Burgdörfer, a theoretical quantum dynamics researcher at the Institute of Theoretical Physics at the Vienna University of Technology, said in a statement.
Put the Rydberg atom in a cold place with other atoms, and it will "swallow" and bond to the other atoms its electrons encircle. The whole assembly — the ultralarge atom and the many atoms stuffed inside — together form a state of matter known as Rydberg polarons. [Wacky Physics: The Coolest Little Particles in Nature]
Enormous atom
To create this weird state of matter, Burgdörfer and his colleagues started with a collection of strontium atoms, which they cooled to just a smidgen above absolute zero, or minus 459.67 degrees Fahrenheit (minus 273.15 degrees Celsius). At this ultralow temperature, the atoms have almost no energy to vibrate or move, and they begin to act as if they were a single atom, becoming a state of matter known as a Bose-Einstein condensate.
From there, the team shined a laser at one of the strontium atoms, jolting it with extra energy. Usually, when electrons jump to a higher energy, they literally leap into an orbit that is farther away from their nucleus. In this case, however, the electron's entire outer orbit swung out, making the atom a Rydberg atom. This monster atom now had a truly gargantuan atomic radius, allowing the electrons to not only circle the atom's nucleus but also engulf many of the other atoms (which were part of the ultracold Bose-Einstein condensate) around the nucleus. When the Bose-Einstein condensate is extremely dense, and the Rydberg atom is highly energized, the enormous Rydberg atom could fit up to 170 strontium atoms in its orbit, the researchers reported Feb. 22 in the journal Physical Review Letters.
But that's not the end of the story. Because the strontium atoms are neutral — meaning their overall charge is zero — they have only the slightest impact on the electrons' path. (The negatively charged electron only generates strong attractive or repulsuve forces with positively or negatively charged particles.)
Get the world’s most fascinating discoveries delivered straight to your inbox.
"The atoms do not carry any electric charge; therefore, they only exert a minimal force on the electron," said study co-author Shuhei Yoshida, also a physicist at the Institute of Theoretical Physics at the Vienna University of Technology.
Instead, as the orbiting electrons veer close to atoms in the condensate, they scatter a bit but do not leave their orbital path. This weak interaction lowers the system's energy — in essence, causing all of the atoms within the Rydberg atom's orbit to bond with the electrons that encircle them.
"It is a highly unusual situation," Yoshida said in the statement. "Normally, we are dealing with charged nuclei, binding electrons around them. Here, we have an electron, binding neutral atoms."
The key is to keep everything very cold; if the atoms in the Bose-Einstein condensate had any more energy to move around, its atoms would break the bonds with the Rydberg atom, the researchers said.
Originally published on Live Science.

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.

 Live Science Plus
Live Science Plus






