Bubbles Get Hotter than the Sun

Just as blowing up a bubble leads to a pop, so can shrinking it. Rapidly collapsing bubbles have long been known to reach astonishing temperatures.
Now scientists have measured just how hot. And they're surprised.
"When bubbles in a liquid get compressed, the insides get hot - very hot," said Ken Suslick of the University of Illinois at Urbana-Champaign. "The temperature we measured - about 20,000 degrees Kelvin [35,540 Fahrenheit] - is four times hotter than the surface of our Sun."
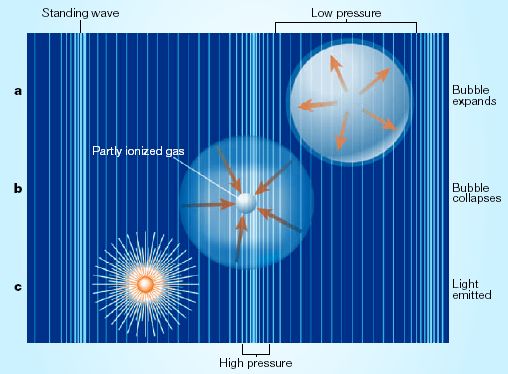
The bubbles are driven to form and collapse in a process called sonoluminescence, in which a liquid is blasted with high-frequency sound waves between 20 and 40 kilohertz (the highest pitch that humans can hear is about 20 kilohertz).
Inside a collapsing bubble, the temperature rises precipitously. Atoms and molecules collide with high-energy particles to create a fourth state of matter, called plasma. The process emits light.
But the heating is so brief and localized that it cannot be measured directly with a thermometer.
The emitted light, however, can be analyzed to determine the temperature of the imploding gas. Previous measurements of multiple-bubble sonoluminescence have found temperatures of 5,000 Kelvin, or 8,500 degrees Fahrenheit.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Suslick and his graduate student David Flannigan were able to measure the temperature of single bubbles, which are not disturbed by neighboring ones. The light from these isolated bubbles is bright enough to be seen by the naked eye.
The high temperatures, measured by Suslick and Flannigan, were partly expected from theory, but solid evidence has been lacking, said Detlef Lohse from the University of Twente, The Netherlands.
The new experiments "are a milestone in single-bubble sonoluminescence, as they constitute the first direct measurement of the temperature and the state of matter in a single bubble at collapse," said Lohse, who was not involved in the work.
The light that is seen is coming from the outer surface of the rapidly shrinking bubble. Inside this surface, the temperature is believed to be even higher. Some have predicted that in these extreme conditions nuclear fusion might occur, but no conclusive evidence has yet been found.
The recent results are reported in the March 3 issue of the journal Nature.
States of Matter
The state of normal matter depends on temperature, which regulates how fast molecules move and therefore how far apart they are. Plasma, a superhot gas, is an entirely different animal, being electrically charged and very expansive.
Plasma is sometimes called a fourth state of matter (in addition to solid, liquid, gas). It's created when gas is superheated and electrons are stripped out, leaving electrically charged particles.
Plasma occurs naturally in interstellar space and in the atmospheres of our Sun and other stars. Scientists also create plasma in labs in order to study emissions from the violent regions around black holes. A fluorescent lamp is an example of a highly contained plasma.
LiveScience graphic, Robert Roy Britt

 Live Science Plus
Live Science Plus






