Small World: Quantum Identity Crisis Observed

A quantum enigma has been put to the test once again, but this time physicists have made the experiment smaller than it has ever been.
The classic double-slit experiment tests the behavior of light, electrons, atoms and some molecules as both particle-like and wave-like, a mysterious duality that has intrigued and puzzled scientists for more than a century.
Light or electrons are aimed at a solid plate with two parallel cuts in it, offering two choices: go through the slit on the left or the slit on the right. Subatomic particles will sometimes break the rules and go through both slits, just as a wave would.
Article continues belowThe most bizarre aspect of this particle-wave duality is that it depends on how much an observer pays attention. The more carefully the observer measures whether it was the left or right slit, the more the object in question chooses a single slit, just as a particle would.
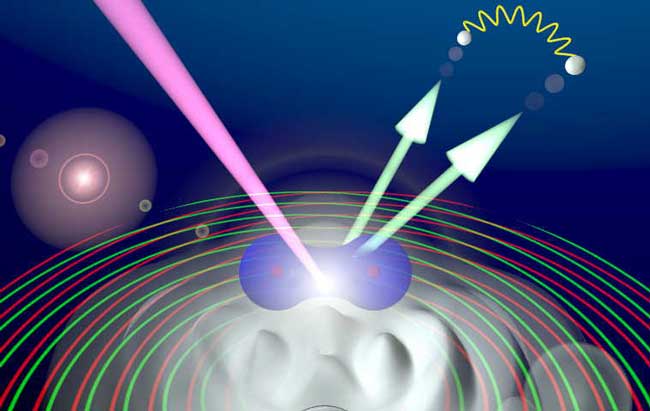
Now an international team of scientists has exhibited this quantum identity crisis using a single hydrogen molecule as their lab equipment.
In this case, an incoming X-ray beam strikes the hydrogen molecule, thereby liberating the two electrons that typically buzz around the molecule's two central nuclei. But before these electrons shoot off into the surrounding space, they make a quick pass by one of the nuclei, which act like left and right slits.
The researchers concentrated on the "fast electron" that carried away the majority of the energy. As expected, the fast electron acted sometimes like a wave and sometimes like a particle.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Interestingly, this behavior depended on the other "slow electron," which interacted very slightly with the fast electron and in so doing played the part of the "observer."
If the slow electron had little energy, it had trouble measuring the fast electron's movement. As a consequence, the fast electron went through both slits like a wave. But if the slow electron had more energy, it got a good look at the fast electron, which responded by choosing one slit like a particle.
The results, detailed in the Nov. 9 issue of the journal Science, give some insight into how a future quantum computer might work as it relies on the phenomena of "superposition" and "entanglement" to perform operations on data stored in units called quantum bits.
- The Enduring Mystery of Light
- The Strangest Little Things in Nature
- Image Gallery: Microscopic Images As Art
