Traditional Chinese Herbal Medicine Yields Its Secrets
This Research in Action article was provided to LiveScience in partnership with the National Science Foundation.
For thousands of years, Chinese herbalists have treated malaria using Chang Shan, a root extract from a type of hydrangea that grows in Tibet and Nepal. Recent studies have suggested Chang Shan can also reduce scar formation, treat multiple sclerosis and even slow cancer progression.
Researchers have suspected that the herbal extract's power to mitigate the effects of malaria stems from febrifugine, the extract's active ingredient. Using halofuginone, a widely studied compound derived from febrifugine, two research teams have helped explain how the extract works. Their findings suggest ways to harness the herbal remedy to treat a host of medical conditions.
Understanding Molecular Changes
To discover the extract's molecular secrets, Harvard School of Dental Medicine researchers and international collaboratorsfocused on themolecular changes triggered by halofuginone. Over the years, they've learned that halofuginone activates a stress-response pathway that blocks the production of a harmful class of immune cells, called Th17 cells. These cells have been implicated in many autoimmune disorders, including inflammatory bowel disease, rheumatoid arthritis and psoriasis.
Most recently, the international team showed that halofuginone restricted the activity of a key enzyme involved in making proteins. Blocking the enzyme kick-started the stress-response pathway, thereby clamping down on the production of TH17 cells and other types of cells involved in inflammation. These findings help explain halofuginone's wide range of therapeutic effects and suggest that the compound could be a useful tool for studying an important molecular process.
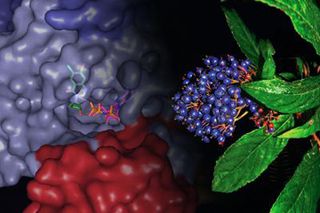
Understanding Molecular Binding
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.
The other study, conducted by scientists at The Scripps Research Institute, focused on how halofuginone binds to its target enzyme. Studies revealed that the compound latches on to and blocks the business end of the enzyme with a "two-handed" grip. In an unusual twist, the researchers discovered that ATP (adenosine triphosphate), a molecule needed for the enzyme to function normally, enables the binding. These details of the herbal compound bound to the enzyme and ATP suggests the medicine's structure could be a useful model in designing drugs to treat numerous other diseases.
Together, these findings point to the power of basic research to reveal new insights into biological processes and new directions for the development of medicines.
This research was supported by the National Institutes of Health. To see more images and videos of basic biomedical research in action, visit NIH's Biomedical Beat Cool Image Gallery.
Editor's Note: Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author and do not necessarily reflect the views of the National Science Foundation. See the Research in Action archive.

